lab notes
Hypothesis
Heat will denature the proteins because it will break the bonds that hold the amino acids together.
Salt will not do anything because it doesn't affect any of the bonds in proteins.
Sodium Bicarbonate will not denature the proteins because although it is a base, it is too weak to disrupt the ionic bonds in the protein.
Lemon juice will denature the proteins because it is a strong acid and it will disrupt the ionic bonds in the protein.
The rubbing alcohol will denature the proteins by breaking the hydrogen bonds present and making new bonds to the amino acids.
Silver nitrate will denature the proteins because it will disrupt the ionic bonds in proteins by exchanging electrons.
Observations
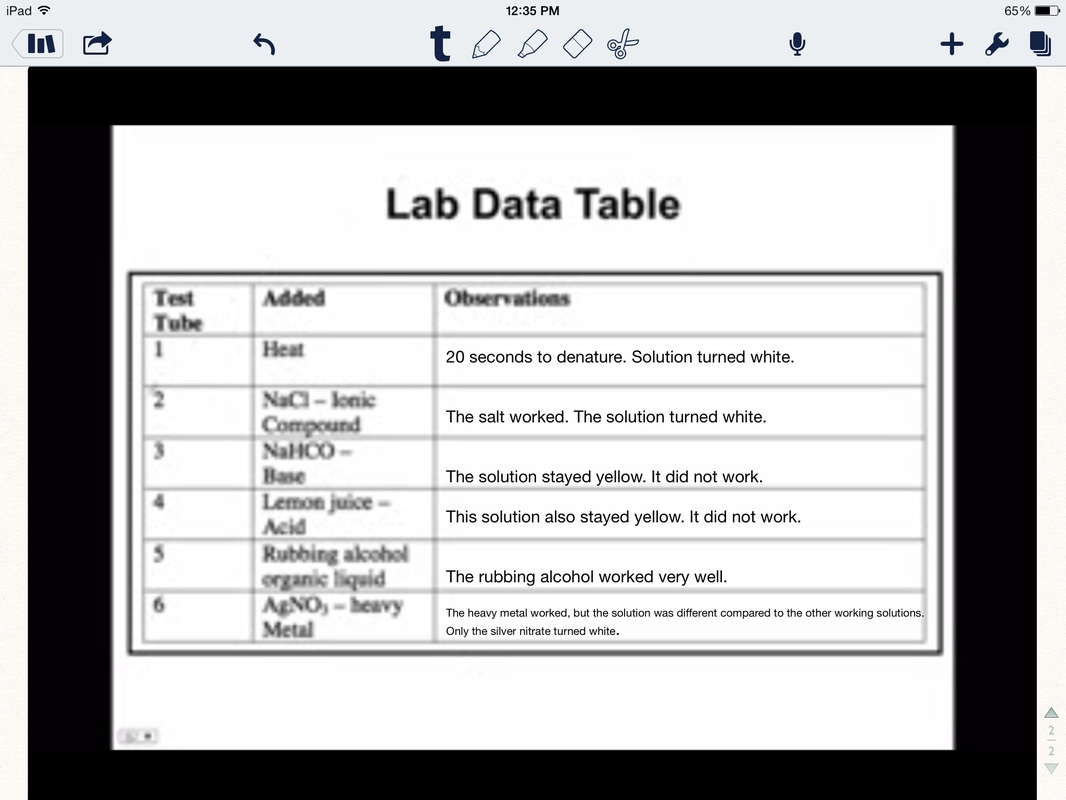
Tube #1: Heat- Time: 20 seconds to denature. Definitely worked. The solution turned white.
Tube #2: Salt- The salt worked. It turned white.
Tube #3: Sodium bicarbonate- It did not work.
Tube #4: Lemon juice- It did not work.
Tube #5: Rubbing alcohol- It did work, turned white.
Tube #6: Silver nitrate- it worked well, it was white. It was different as only the parts the silver nitrate touched turned white while the rest of the egg material stayed in its natural yellowish color.
Post-Lab Questions
Which method appeared to have the most dramatic denaturing effect on egg albumin? Why do you think this method had the biggest greatest effect? The rubbing alcohol was the method that worked the best. The egg white turned white the fastest and also was the whitest of any of the solution when it was mixed with the rubbing alcohol. It probably worked the best because it was very strong alcohol and was very effective at breaking the bonds.
Of the methods you tested, which would be most likely to be used in the food industry? The method that would have been most likely used in denaturing proteins in the food industry is heat. Heat is the most common way of denaturing proteins when cooking, such as cooking steak and eggs because it adds nothing to the food being cooked like the other methods.
Heat will denature the proteins because it will break the bonds that hold the amino acids together.
Salt will not do anything because it doesn't affect any of the bonds in proteins.
Sodium Bicarbonate will not denature the proteins because although it is a base, it is too weak to disrupt the ionic bonds in the protein.
Lemon juice will denature the proteins because it is a strong acid and it will disrupt the ionic bonds in the protein.
The rubbing alcohol will denature the proteins by breaking the hydrogen bonds present and making new bonds to the amino acids.
Silver nitrate will denature the proteins because it will disrupt the ionic bonds in proteins by exchanging electrons.
Observations
Tube #1: Heat- Time: 20 seconds to denature. Definitely worked. The solution turned white.
Tube #2: Salt- The salt worked. It turned white.
Tube #3: Sodium bicarbonate- It did not work.
Tube #4: Lemon juice- It did not work.
Tube #5: Rubbing alcohol- It did work, turned white.
Tube #6: Silver nitrate- it worked well, it was white. It was different as only the parts the silver nitrate touched turned white while the rest of the egg material stayed in its natural yellowish color.
Post-Lab Questions
Which method appeared to have the most dramatic denaturing effect on egg albumin? Why do you think this method had the biggest greatest effect? The rubbing alcohol was the method that worked the best. The egg white turned white the fastest and also was the whitest of any of the solution when it was mixed with the rubbing alcohol. It probably worked the best because it was very strong alcohol and was very effective at breaking the bonds.
Of the methods you tested, which would be most likely to be used in the food industry? The method that would have been most likely used in denaturing proteins in the food industry is heat. Heat is the most common way of denaturing proteins when cooking, such as cooking steak and eggs because it adds nothing to the food being cooked like the other methods.
Essay on lab
Michael Tellini, Calvin Foss, Christopher Potter, and Alex Davies
Mr. Helms
Biology 1 (Period D)
12/11/13
Before our group started the lab, we needed to know more. First we must ask ourselves, what is a protein? What does it mean to denature one? We did research to find this out. A protein is an organic compound made up of amino acids. These acids are held together by bonds. Denaturing is the process in which the natural qualities of something are taken away. When the bonds are broken by a chemical or substance, the amino acids fall apart and the proteins can not function anymore. That is what happens when proteins are denatured.
Before we started the lab where we denatured the proteins in egg whites, we came up with hypotheses for each method of denaturing. We predicted heat would denature the proteins because it would break the bonds that held the amino acids together. We predicted salt would not do anything because it wouldn't affect any of the bonds in proteins. We predicted sodium bicarbonate would not denature the proteins because although it is a base, it would be too weak to disrupt the ionic bonds in the protein. We predicted lemon juice would denature the proteins because it is a strong acid and it would disrupt the ionic bonds in the protein. We predicted the rubbing alcohol would denature the proteins by breaking the hydrogen bonds present and making new bonds to the amino acids. Finally, we predicted silver nitrate would denature the proteins because it would disrupt the ionic bonds in proteins by exchanging electrons.
During the lab, we went through an assortment of steps to attempt to denature the proteins in each of the vials. The first step we took was to fill each vial halfway full with egg white. We had six different vials that we tested differently to see whether or not certain methods work to denature proteins. The first method we tested was heat. We filled a 400 ml beaker 200 ml full with water and boiled the water using gas. After the water was boiling, we inserted the closed vial and waited for the egg white to turn white instead of its normal yellowish color. After twenty seconds the egg yoke turned white. With the second vial, we inserted salt to the egg white. Upon shaking the vial, the egg white quickly turned white. The third method we tested involved sodium bicarbonate. The solution did not turn white, meaning the proteins did not denature. The fourth method we tested was lemon juice, which did not denature the proteins either. The fifth method we used was putting rubbing alcohol in the solution. We shook the vial, and found that the new solution turned whiter than any other. The last method we used was silver nitrate, which had an interesting effect. The part of the solution that was touched by the silver nitrate turned white, but the rest stayed in its natural yellowish color. Even upon shaking the vial, we found that the solution was only white in certain areas of the vial. The conclusion we came to was that the silver nitrate worked, but only slightly, and could not spread. The data table shown below shows our findings on which methods we found denatured the proteins. The hypothesis that we constructed was right in some areas, and wrong in others. We predicted that salt would have no effect, but it did. We were also wrong on predicting that lemon juice would denature the proteins, as it did not denature them. We were right though on the majority of our hypothesis. The lab taught us that protein denaturation is essential to the building of new proteins, but can only be done by using certain methods.
The denaturation of proteins is most effective when rubbing alcohol interacts with proteins. We were able to conclude this by mixing one tablespoon of rubbing alcohol with half a flask of raw albumen which resulted in it turning white, which meant the proteins were denatured. The fact that the alcohol seemed to denature the fastest and make it most white led us to believe the most effective protein denaturation substance is alcohol. The data we observed with all the vials showed the substances that can and cannot react to the denaturation of proteins.
This lab was a very fascinating way to see how protein denaturation takes place. Prior to the lab, we knew little about the breaking down of proteins and the building of new ones. We now know that the denaturation of proteins is what changes the purpose and function of those proteins. The lab showed that many different methods can be used to denature proteins. The most effective was rubbing alcohol, although heat is perhaps the safest if somebody is consuming the denatured proteins as in the food industry. From our results, we saw that heat, salt, rubbing alcohol, and silver nitrate denatured the proteins while lemon juice and sodium bicarbonate did not.
Mr. Helms
Biology 1 (Period D)
12/11/13
Before our group started the lab, we needed to know more. First we must ask ourselves, what is a protein? What does it mean to denature one? We did research to find this out. A protein is an organic compound made up of amino acids. These acids are held together by bonds. Denaturing is the process in which the natural qualities of something are taken away. When the bonds are broken by a chemical or substance, the amino acids fall apart and the proteins can not function anymore. That is what happens when proteins are denatured.
Before we started the lab where we denatured the proteins in egg whites, we came up with hypotheses for each method of denaturing. We predicted heat would denature the proteins because it would break the bonds that held the amino acids together. We predicted salt would not do anything because it wouldn't affect any of the bonds in proteins. We predicted sodium bicarbonate would not denature the proteins because although it is a base, it would be too weak to disrupt the ionic bonds in the protein. We predicted lemon juice would denature the proteins because it is a strong acid and it would disrupt the ionic bonds in the protein. We predicted the rubbing alcohol would denature the proteins by breaking the hydrogen bonds present and making new bonds to the amino acids. Finally, we predicted silver nitrate would denature the proteins because it would disrupt the ionic bonds in proteins by exchanging electrons.
During the lab, we went through an assortment of steps to attempt to denature the proteins in each of the vials. The first step we took was to fill each vial halfway full with egg white. We had six different vials that we tested differently to see whether or not certain methods work to denature proteins. The first method we tested was heat. We filled a 400 ml beaker 200 ml full with water and boiled the water using gas. After the water was boiling, we inserted the closed vial and waited for the egg white to turn white instead of its normal yellowish color. After twenty seconds the egg yoke turned white. With the second vial, we inserted salt to the egg white. Upon shaking the vial, the egg white quickly turned white. The third method we tested involved sodium bicarbonate. The solution did not turn white, meaning the proteins did not denature. The fourth method we tested was lemon juice, which did not denature the proteins either. The fifth method we used was putting rubbing alcohol in the solution. We shook the vial, and found that the new solution turned whiter than any other. The last method we used was silver nitrate, which had an interesting effect. The part of the solution that was touched by the silver nitrate turned white, but the rest stayed in its natural yellowish color. Even upon shaking the vial, we found that the solution was only white in certain areas of the vial. The conclusion we came to was that the silver nitrate worked, but only slightly, and could not spread. The data table shown below shows our findings on which methods we found denatured the proteins. The hypothesis that we constructed was right in some areas, and wrong in others. We predicted that salt would have no effect, but it did. We were also wrong on predicting that lemon juice would denature the proteins, as it did not denature them. We were right though on the majority of our hypothesis. The lab taught us that protein denaturation is essential to the building of new proteins, but can only be done by using certain methods.
The denaturation of proteins is most effective when rubbing alcohol interacts with proteins. We were able to conclude this by mixing one tablespoon of rubbing alcohol with half a flask of raw albumen which resulted in it turning white, which meant the proteins were denatured. The fact that the alcohol seemed to denature the fastest and make it most white led us to believe the most effective protein denaturation substance is alcohol. The data we observed with all the vials showed the substances that can and cannot react to the denaturation of proteins.
This lab was a very fascinating way to see how protein denaturation takes place. Prior to the lab, we knew little about the breaking down of proteins and the building of new ones. We now know that the denaturation of proteins is what changes the purpose and function of those proteins. The lab showed that many different methods can be used to denature proteins. The most effective was rubbing alcohol, although heat is perhaps the safest if somebody is consuming the denatured proteins as in the food industry. From our results, we saw that heat, salt, rubbing alcohol, and silver nitrate denatured the proteins while lemon juice and sodium bicarbonate did not.